JOURNEY II Active Knee System
JOURNEY◊ II XR
Rediscover your active life with the high performance
A New Chapter in Knee Implants
Recent advances in biomedical engineering software have opened a new chapter in high performance knee implants.
One remarkable breakthrough has been the creation of the JOURNEY◊ II XR Active Knee System, which combines an implant designed to restore the stability and natural motion of the human knee with low-friction materials that may help extend the longevity of the implant itself.
While the longevity of a knee implant is heavily influenced by the materials used to make it, the natural feeling of the implant during physical activity is dependent upon the way the patient's muscles, ligaments and tendons are addressed during surgery and by the implant's shape within the body after surgery.
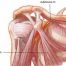
Every knee implant design is faced with two important challenges; how to recreate the normal swing-and-rotate motion of your natural knee, and how to restore stability to your joint after surgery.
For the first challenge, conventional knee implant designs have attempted to recreate the natural, fluid motion of the knee with either a rotating platform (a simple pivot point) within the implant or by requiring an angled alignment of the implant during surgery. Both of these options force the muscles around your knew joint to work harder as they adjust to the joint's new and unnatural shapes and movement patterns.
For the second challenge, most implants rely on design aspects within the plastic insert to provide the stability that is sacrificed after the anterior and posterior cruciate ligaments (ACL/PCL) within your joint are removed to accommodate the new implant.
JOURNEY II XR is Different
Fortunately, the JOURNEY II XR Knee Implant is different from most knee implants.
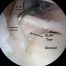
First, the JOURNEY II XR implant features an anatomically shaped femoral component and two plastic inserts designed to work together to reproduce the original internal shapes and forces of your natural knee as it goes through its full range of motion. This attention to anatomic detail means that the muscles and other soft tissues around your joint don't have to adjust to unfamiliar stresses and you may be able to return to a natural pattern of motion after surgery.
Second, the JOURNEY II XR uses a U-shaped tibial base plate that fits around your healthy ACL and PCL, allowing them to remain in place and function normally after surgery. In short, the function of these important ligaments doesn't have to be re-engineered because they were never removed.
Added Durability
The JOURNEY II XR implant addresses durability with VERILAST◊ Technology, a combination of two wear reducing materials - the proprietary OXINIUM◊ metal alloy and a highly cross-linked plastic insert - that were designed to address wear on both surfaces of the implant.
Because it is twice as hard as cobalt chrome, the most commonly used metal in knee implants, implants made with OXINIUM material have been shown in lab testing to reduce joint wear by more than 80% when compared to cobalt chromium components.1
VERILAST Technology was designed to address "wear and tear," which is only one of several reasons why a knee implant may need to be replaced. Each patient should listen carefully when his or her orthopedic surgeon reviews other risks that can shorten the life of a new knee - such as infection, weight gain or high impact sports.
The results of laboratory wear simulation testing have not been proven to predict actual joint durability and performance in people. A reduction in wear alone may not result in improved joint durability and performance because other factors can affect joint durability and performance and cause medical conditions that may result in the need for additional surgery. These other factors were not studied as part of the testing.
Not all patients are candidates for the JOURNEY II XR Knee Implant. Discuss your condition and implant options with your surgeon.
Disclaimer
Individual results of joint replacement vary. Implants are intended to relieve knee pain and improve function, but may not produce the same feel or function as your original knee. There are potential risks with knee replacement surgery such as loosening, wear and infection that may result in the need for additional surgery. Patients should not perform high impact activities such as running and jumping unless their surgeon tells them that the bone has healed and these activities are acceptable. Early device failure, breakage or loosening may occur if a surgeon's limitations on activity level are not followed.
References
- jHunter, G., and Long, M., Abrasive Wear of Oxidized Zr-2.5Nb, CoCrMo, and Ti-6Al-4V Against Bone Cement. 6th World Biomaterials, Minneapolis, MN 2000, p. 835
Dr. Philip Schmitt
Offers JOURNEY II Active Knee System
Bone and Joint Specialists Orthopaedic Hip Surgeon Dr. Philip Schmitt specializes in the treatment of knee injuries and knee disorders, including arthritis and trauma. He understands the importance of getting you back to an Active Lifestyle.
Dr. Schmitt can evaluate and diagnose your knee pain injuries and conditions, and provide a customized treatment plan that is exclusive to you. He will explore conservative nonsurgical methods first, such as physical therapy, including individualized knee exercises to relieve your pain. However, if surgery is necessary to allow you to continue an active lifestyle, Dr. Schmitt will employ specialized techniques that are minimally invasive and can lead to quicker recovery times.